1. Subatomic Particles
- Atoms are mostly empty space surrounding a very small, dense nucleus that contains protons and neutrons.
- Electrons are found in shells in the empty space around the nucleus
Properties of Subatomic Particles
| Particle | Relative Mass | Relative Charge | Location |
|---|---|---|---|
| Proton ( | Nucleus | ||
| Neutron ( | Nucleus | ||
| Electron ( | Shells |
2. Definitions and Notation
Standard Nuclide Notation:
- Atomic Number (
): The number of protons in the nucleus. - Mass Number (
): The total number of protons + neutrons in the nucleus.
Calculation Example: Ion
- Protons: 34 (from periodic table).
- Neutrons:
. - Electrons:
(Anion has gained electrons).
3. Behaviour in Electric Fields
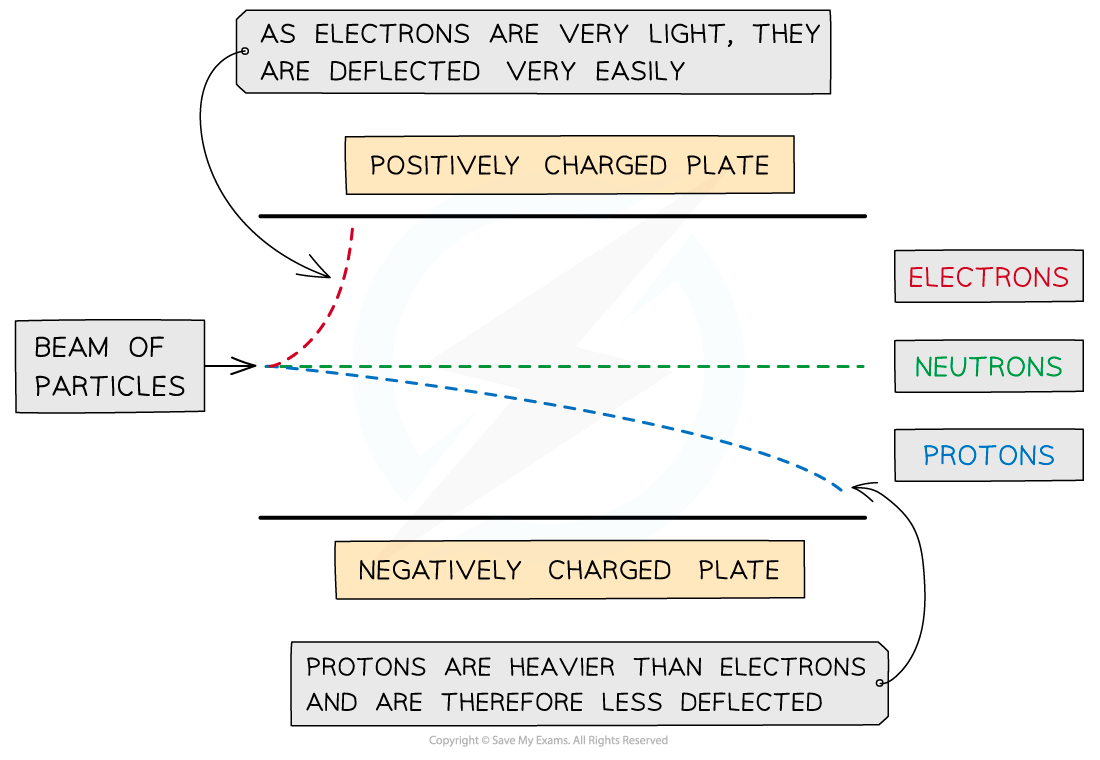
When beams of subatomic particles move at the same velocity through an electric field, their deflection depends on their Charge-to-Mass ratio (
Deflection Summary
| Beam | Direction of Deflection | Magnitude of Deflection | Reason |
|---|---|---|---|
| Protons | Towards negative plate | Small | High mass ( |
| Electrons | Towards positive plate | Very Large | Very low mass ( |
| Neutrons | No deflection | None | Charge is |
4. Atomic and Ionic Radius Trends
Atomic Radius
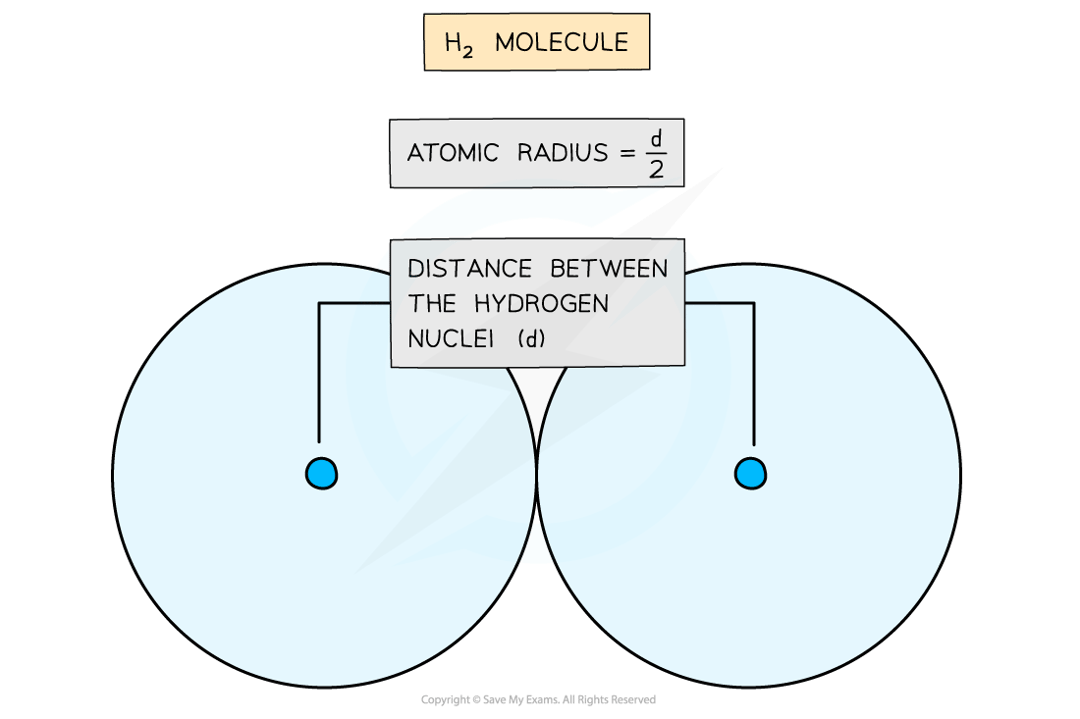
It is half the distance between the two nuclei of two covalently bonded atoms of the same type.
The atomic radius of a hydrogen atom
A. Atomic Radius Across a period (e.g. Na
Explanation
Across a period:
- The nuclear charge increases.
- Electrons are added to the same outer shell.
- Shielding is relatively unchanged.
- There is a greater attraction between the nucleus and the outer electrons, pulling the electrons closer to the nucleus.
B. Atomic Radius Down a group
Explanation
Down a group:
- The number of electron shells increases.
- The distance between the nucleus and outer electrons increases.
- Shielding increases.
- The nuclear attractions for outer electrons decreases.
C. Ionic Radius: Cation vs Parent Atom (e.g.
Explanation
- The atom loses its outer shell electrons (one fewer shell).
- There is less electron-electron repulsion.
- The remaining electrons are attracted by the same nuclear charge and are pulled closer to the nucleus
D. Ionic Radius: Anion vs Parent Atom (e.g.
Explanation
- The atom gains electrons into the same outer shell.
- This causes increased electron-electron repulsion.
- The same nuclear charge must hold more electrons, so the electrons move further apart.
E. Ionic Radius Trend Across a Period (The “Step Change”)
Ionic radius does not decrease continuously across a period. There is a distinct “jump” between the group 14 and 15 ions (e.g., Period 3).
Visualizing Period 3:
- Cations (
to ): Radius decreases (Isoelectronic with ). - The Jump (
to ): Radius increases dramatically. - Anions (
to ): Radius decreases (Isoelectronic with ).
Explanation of the Jump
Why is
much larger than ?
- Shells: The cations have lost their outer shell (occupying
). The anions have gained electrons to fill the next shell ( ). - Shielding: The anions have an extra electron shell, significantly increasing shielding and distance from the nucleus compared to the cations.
5. Isoelectronic Series
Isoelectronic species have the same number of electrons and the same electronic configuration.
Example:
Explanation: Why is
smaller than
- They have the same number of electrons and same shielding.
has a greater nuclear charge than . - There is a greater nuclear attraction for the remaining electrons in
, pulling them closer to the nucleus.