What is Solubility?
In A Level chemistry, we deal with “sparingly soluble” salts (salts that barely dissolve, like Silver Chloride or Barium Sulfate). Even though they look insoluble, a tiny amount does dissolve to form a saturated solution.
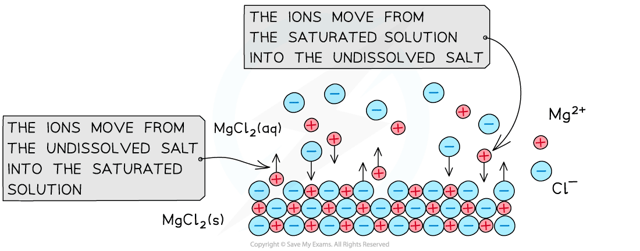
When an undissolved ionic compound is in contact with a saturated solution of its ions, an equilibrium is established:
Solubility Product
Consider the equilibrium when the sparingly soluble salt
The equilibrium constant (
Since the concentration of the solid
Definition: Solubility Product (
) The Solubility Product (
) is:
- The product of the concentrations of each ion in a saturated solution of a sparingly soluble salt
- At 298 K
- Raised to the power of their relative concentrations.
Ionic Product
The Ionic Product (
Important
: Describes the maximum possible concentration (Saturation limit). : Describes the current concentration.
Predicting Precipitation
By comparing the value of
| Condition | State of Solution | Physical Result |
|---|---|---|
| Unsaturated | No precipitate forms. More solid can dissolve. | |
| Saturated | No precipitate forms, but no more solid can dissolve. Equilibrium is formed. | |
| Supersaturated | Precipitate forms. The excess ions crash out of solution until | |
| the quick brown fox jumps over the lazy dog |